


UNTERSTÜTZUNG FÜR IHRE BIOTECH-REISE
MIT PAN-BIOTECH GMBH
PAN-Biotech baut seine Fähigkeiten und sein Produktangebot in den folgenden Schlüsselbereichen aktiv aus:

ZELL- UND GENTHERAPIE:
Entwicklung spezieller Medien und Reagenzien zur Unterstützung des Wachstums und der Produktion von Zellen und Genen für therapeutische Anwendungen, wodurch die Wirksamkeit und Sicherheit modernster Behandlungen verbessert wird
3

IMPFSTOFFPRODUKTION:
Expansion in Technologien zur Impfstoffproduktion, um die notwendigen Werkzeuge und Lösungen für die schnelle und effiziente Entwicklung von Impfstoffen bereitzustellen, auch solchen gegen neu auftretende Krankheiten.
3

mRNA-TECHNOLOGIE:
Innovationen in der mRNA-Technologie, einschließlich der Entwicklung optimierter Medien und Verabreichungssysteme, um den wachsenden Bereich der mRNA-basierten Therapeutika und Impfstoffe zu unterstützen.
3

ANTIKÖRPERPRODUKTION:
Verbesserung von Produkten und Prozessen zur Herstellung hochwertiger Antikörper für Forschung, Diagnostik und therapeutische Anwendungen.
3
Unter der Leitung von Präsident und CEO Jens Hartmann setzt PAN-Biotech seinen Erfolg fort und lässt sich dabei von Grundwerten leiten, die Innovation, Integrität und Kundenorientierung betonen.
„Bei PAN-Biotech werden wir von unserer Leidenschaft für die Wissenschaft und unserem Engagement für Exzellenz angetrieben. Unsere Mission ist es, unsere Kunden mit den Werkzeugen auszustatten, die sie benötigen, um einen bedeutenden Einfluss auf die Welt zu nehmen.“
Mit diesen Werten, die uns leiten, ist PAN-Biotech gut aufgestellt, um in der Biotechnologiebranche führend zu sein und die Zukunft von Wissenschaft und Gesundheitswesen zu gestalten.

INNOVATION:
PAN-Biotech investiert massiv in Forschung und Entwicklung und fördert eine Kultur der Kreativität und wissenschaftlichen Forschung. Das Unternehmen ermutigt seine Teams, neue Ideen zu erforschen und die Grenzen des Möglichen in der Biotechnologie zu erweitern.
3

INTEGRITÄT:
Ethische Praktiken und Transparenz stehen im Mittelpunkt der Geschäftstätigkeit von PAN-Biotech. Das Unternehmen verpflichtet sich zu ehrlichem und verantwortungsvollem Handeln und baut Vertrauen bei Kunden, Partnern und der wissenschaftlichen Gemeinschaft auf.
3

KUNDENORIENTIERUNG:
PAN-Biotech ist bestrebt, die spezifischen Bedürfnisse seiner Kunden zu verstehen und zu erfüllen. Durch personalisierte Lösungen und herausragenden Kundenservice stellt das Unternehmen sicher, dass seine Produkte und Dienstleistungen den Zielen und Vorgaben seiner Kunden entsprechen.
3

QUALITÄT:
Ein Hauptaugenmerk liegt auf der Lieferung von Spitzenprodukten „Made in Germany“ nach höchsten Standards, um sicherzustellen, dass jedes Produkt den Anforderungen an Exzellenz und Zuverlässigkeit entspricht und unser Engagement für höchste Qualität widerspiegelt.
3
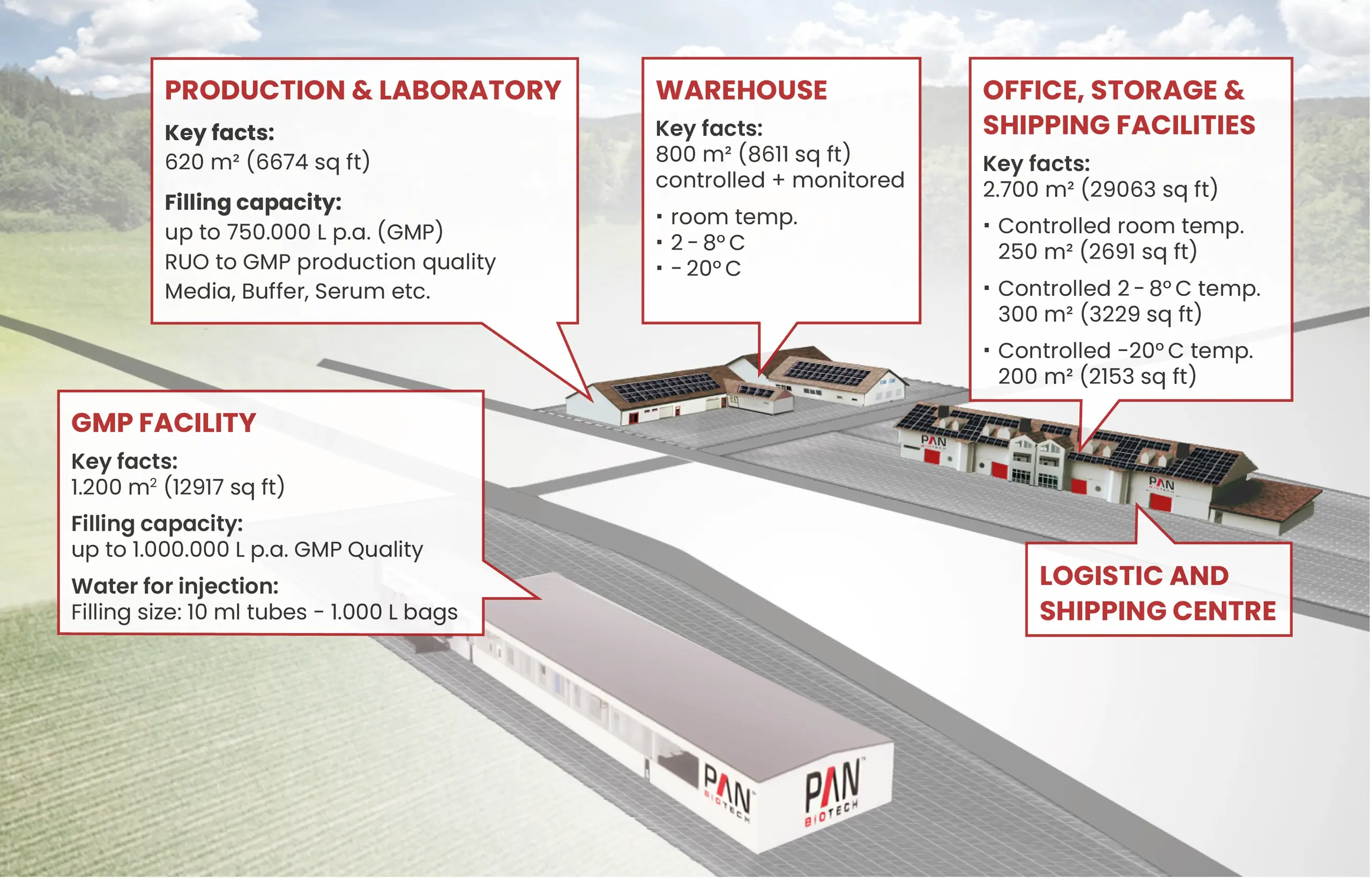
ENTDECKEN SIE UNSER PAN-VILLAGE
You are currently viewing a placeholder content from Default. To access the actual content, click the button below. Please note that doing so will share data with third-party providers.
More InformationKOMMEN SIE UNS BESUCHEN
CPHI Südostasien 2025
Malaysia International Trade & Convention Center, Kuala Lumpur - Treffen Sie uns im Bayerischen Pavillon, Stand J16-5


